PRODUCT IDENTIFICATION
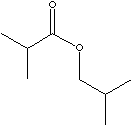
2-Methylpropyl isobutyrate; Isobutyl isobutyrate; IBIB;
2-Methyl-1-propyl 2-methylpropanoate; 2-Methylpropyl 2-methylpropanoate; 2-Methylpropyl 2-methylpropionate; 2-Methylpropyl isobutyrate; Isobutyl 2-methylpropanoate; Isobutyl isobutanoate; Isobutylester kyseliny isomaselne; Isobutyric acid isobutyl ester; 2-mMthylpropanoic acid 2-methylpropyl ester;
CLASSIFICATION
EXTRA NOTES
UN2528 [Flammable liquid]
PHYSICAL AND CHEMICAL PROPERTIES
clear liquid
0.856
2.68
REFRACTIVE INDEX
Health: 2; Flammability: 2; Instability: 0
40 C
EXTERNAL LINKS & GENERAL DESCRIPTION
Wikipedia Linking - Ester
Drug Information Portal (U.S. National Library of Medicine) - Isobutyl isobutyrate
PubChem Compound Summary - Isobutyl isobutyrate
KEGG (Kyoto Encyclopedia of Genes and Genomes) - Isobutyl isobutyrate
http://www.ebi.ac.uk/chebi/ - Isobutyl isobutyrate
http://www.ncbi.nlm.nih.gov/ - Isobutyl isobutyrate
Human Metabolome Database - Isobutyl isobutyrate
http://www.thegoodscentscompany.com/
Local:
Carboxylic acid
is an organic compound whose molecules contain carboxyl group and have the
condensed chemical formula R-C(=O)-OH in which a carbon atom is bonded to an
oxygen atom by a solid bond and to a hydroxyl group by a single bond), where R
is a hydrogen atom, an alkyl group, or an aryl group. Carboxylic acids can be
synthesized if aldehyde is oxidized. Aldehyde can be obtained by oxidation of
primary alcohol. Accordingly, carboxylic acid can be obtained by complete
oxidation of primary alcohol. A variety of Carboxylic acids are abundant in
nature and many carboxylic acids have their own trivial names. Examples are
shown in table. In substitutive nomenclature, their names are formed by adding
-oic acid' as the suffix to the name of the parent compound. The first character
of carboxylic acid is acidity due to dissociation into H+ cations and
RCOO- anions in aqueous
solution. The two oxygen atoms are electronegatively charged and the hydrogen of
a carboxyl group can be easily removed. The presence of electronegative groups
next to the carboxylic group increases the acidity. For example, trichloroacetic
acid is a stronger acid than acetic acid. Carboxylic acid is useful as a parent
material to prepare many chemical derivatives due to the weak acidity of the
hydroxyl hydrogen or due to the difference in electronegativity between carbon
and oxygen. The easy dissociation of the hydroxyl oxygen-hydrogen provide
reactions to form an ester with an alcohol and to form a water-soluble salt with
an alkali. Almost infinite esters are formed through condensation reaction
called esterification between carboxylic acid and alcohol, which produces water.
The second reaction theory is the addition of electrons to the
electron-deficient carbon atom of the carboxyl group. One more theory is
decarboxylation (removal of carbon dioxide form carboxyl group). Carboxylic
acids are used to synthesize acyl halides and acid anhydrides which are
generally not target compounds. They are used as intermediates for the synthesis
esters and amides, important derivatives from carboxylic acid in biochemistry as
well as in industrial fields. There are almost infinite esters obtained from
carboxylic acids. Esters are formed by removal of water from an acid and an
alcohol. Carboxylic acid esters are used as in a variety of direct and indirect
applications. Lower chain esters are used as flavouring base materials,
plasticizers, solvent carriers and coupling agents. Higher chain compounds are
used as components in metalworking fluids, surfactants, lubricants, detergents,
oiling agents, emulsifiers, wetting agents textile treatments and emollients,
They are also used as intermediates for the manufacture of a variety of target
compounds. The almost infinite esters provide a wide range of viscosity,
specific gravity, vapor pressure, boiling point, and other physical and chemical
properties for the proper application selections. Amides are formed from the
reaction of a carboxylic acids with an amine. Carboxylic acid's reaction to link
amino acids is wide in nature to form proteins (amide), the principal
constituents of the protoplasm of all cells. Polyamide is a polymer containing
repeated amide groups such as various kinds of nylon and polyacrylamides.
Carboxylic acid are in our lives.
|
ALIPHATIC CARBOXYLIC ACIDS | ||||
|
COMMON NAME |
SYSTEMATIC NAME |
CAS RN |
FORMULA |
MELTING POINT |
|
Formic Acid |
Methanoic acid | 64-18-6 |
HCOOH |
8.5 C |
| Acetic Acid | Ethanoic acid | 64-19-7 | CH3COOH |
16.5 C |
| Carboxyethane | Propionic Acid | 79-09-4 | CH3CH2COOH |
-21.5 C |
| Butyric Acid | n-Butanoic acid | 107-92-6 | CH3(CH2)2COOH |
-8 C |
| Valeric Acid | n-Pentanoic Acid | 109-52-4 | CH3(CH2)3COOH |
-19 C |
| Caproic Acid | n-Hexanoic Acid | 142-62-1 | CH3(CH2)4COOH |
-3 C |
| Enanthoic Acid | n-Heptanoic acid | 111-14-8 | CH3(CH2)5COOH |
-10.5 C |
| Caprylic Acid | n-Octanoic Acid | 124-07-2 | CH3(CH2)6COOH |
16 C |
| alpha-Ethylcaproic Acid | 2-Ethylhexanoic Acid | 149-57-5 | CH3(CH2)3CH(C2H5)COOH |
-59 C |
| Valproic Acid | 2-Propylpentanoic Acid | 99-66-1 | (CH3CH2CH2)2CHCOOH |
120 C |
| Pelargonic Acid | n-Nonanoic Acid | 112-05-0 | CH3(CH2)7COOH |
48 C |
| Capric Acid | n-Decanoic Acid | 334-48-5 | CH3(CH2)8COOH |
31 C |
There are almost infinite esters obtained from thousands of potential starting materials. Esters are formed by removal of water from an acid and an alcohol, e.g., carboxylic acid esters, phosphoric acid esters, and sulfonic acid esters. Carboxylic acid esters are used as in a variety of direct and indirect applications. Lower chain esters are used as flavouring base materials, plasticizers, solvent carriers and coupling agents. Higher chain compounds are used as components in metalworking fluids, surfactants, lubricants, detergents, oiling agents, emulsifiers, wetting agents textile treatments and emollients, They are also used as intermediates for the manufacture of a variety of target compounds. The almost infinite esters provide a wide range of viscosity, specific gravity, vapor pressure, boiling point, and other physical and chemical properties for the proper application selections.
APPEARANCE
clear liquid
PURITY
98.0% min
ACIDITY
1.0 max
REFRACTIVE INDEX
SPECIFIC GRAVITY
0.845 - 0.865
HAZARD OVERVIEW
Flammable liquid and vapor. Causes eye, skin, and respiratory tract irritation. Target Organs: Central nervous system, respiratory system, eyes, skin. OSHA Hazards:Combustible Liquid, Irritant
GHS
PICTOGRAMS


HAZARD STATEMENTS
H226 H315 H319 H335
P STATEMENTS
P210 P261 P280 P302+P352+P338
Xi Irritant
RISK PHRASES
10-36/37/38
SAFETY PHRASES
16-26-36
PRICE INFORMATION